[ad_1]
Whereas Covishield and Covaxin lead the Covid-19 vaccination effort in India, different vaccines, too, are below growth or stay unused after approval from the Centre
Whereas Covishield and Covaxin lead the Covid-19 vaccination effort in India, different vaccines, too, are below growth or stay unused after approval from the Centre
As India nears the 200-crore COVID-19 vaccination mark, Serum Institute of India’s (SII) Covishield accounts for 80 per cent (154 crore) of the jabs, whereas Bharat Biotech’s Covaxin accounts for 16.75 per cent (32 crore) jabs.
Of the 191 crore jabs administered until now, the remaining three vaccines used — Gamelaya’s Sputnik –V, Organic E’s Corbevax and Novovax-SII’s Covovax — account just for 3.25 per cent (6.25 crore) altogether.
As of date, India has granted emergency utilization authorisation (EUA) to 9 COVID-19 vaccine candidates in whole – Covishield, Covaxin, Covovax, Corbevax, Zydus Cadila’s ZyCoV-D, Moderna’s Spikevax, Sputnik-V, Sputnik Mild, and Johnson & Johnson’s (J&J) Ad26.COV2.S.
Nevertheless, solely 5 vaccines are in use and all are indigenously produced.
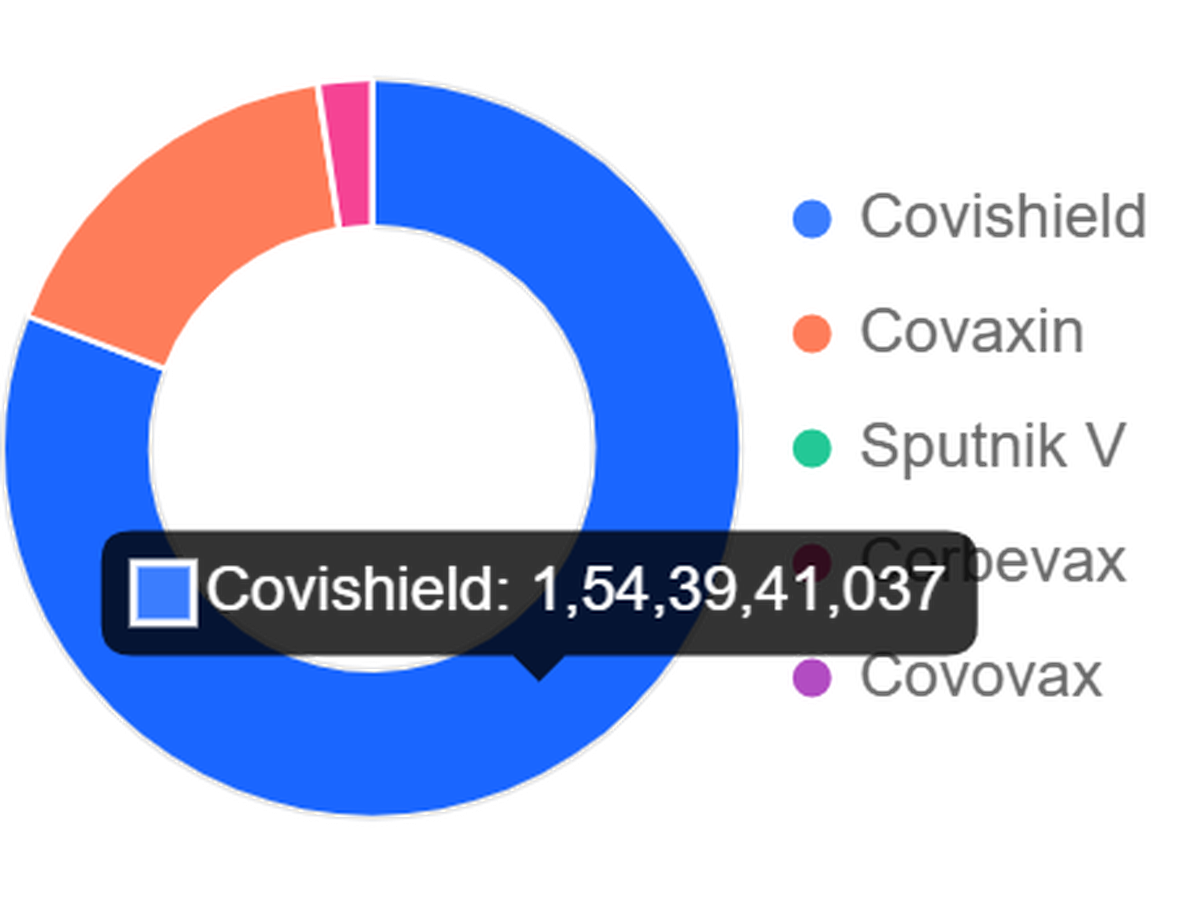
Vaccines in use in India
| Photograph Credit score: –
The 4 COVID-19 vaccines accepted however not in use in India
ZyCoV-D:
Produced by Ahmedabad-based Zydus Cadila in collaboration with the Centre’s Nationwide Biopharma Mission, ZyCoV-D is India’s first DNA COVID-19 vaccine. Utilizing a plasmid DNA — a small, round and extrachromosomal bacterial DNA — ZyCoV-D creates spike protein to develop anti-bodies to counter the COVID-19 virus. Injected in three doses with a disposable painless jet applicator at an interval of 28 days every, ZyCoV-D clocked a vaccine efficacy of 66.6 per cent.
Whereas the Drug Controller Basic of India (DCGI) granted approval to ZyCoV-D on August 20, 2021, the producer and the Centre couldn’t agree on the value for every dose. Zydus Cadila had quoted a worth of Rs 1900 for its three-dose routine and its jet applicators used for every dose. After a collection of negotiations, the corporate diminished its price to Rs 265 per dose and Rs 93 for the applicator – tallying to Rs 358 per dose (Rs 1074 for 3 doses).
Lastly, in February 2022, Zydus Cadila began supplying its COVID-19 vaccine to the Centre, which determined to introduce it in Bihar, Jharkhand, Maharashtra, Punjab, Tamil Nadu, Uttar Pradesh and West Bengal. These states have been requested to establish districts with a excessive quantity of people that haven’t taken the primary dose of the COVID-19 vaccine. Nevertheless, in accordance with the CoWIN Portal, not a single dose of ZyCoV-D has been administered outdoors of medical trials till now.
Pfizer:
Regardless of prolonged discussions, U.S. pharma large Pfizer and the Central authorities couldn’t agree on beneficial phrases for vaccine distribution in India. The Pfizer-BioNTech mRNA-based Covid-19 vaccine, offered below the model title Comirnaty, is among the many most predominantly used vaccines in Europe and the U.S.
Whereas the Centre had fast-tracked clearances for importing international COVID-19 vaccines to India in June 2021, the U.S. pharma firm had locked horns with the Centre over the indemnity clause. Pfizer insisted on an indemnity clause which might defend the corporate if its vaccine was perceived to have brought on demise or any lasting harm to a recipient. Beneath such a clause, any compensation declare must be paid by the federal government, not the corporate.
Hesitating to comply with this binding clause, Centre held a number of rounds of talks with Pfizer to barter costs and the quantity of a possible vaccine order. Pfizer had additionally utilized for EUA in India however later withdrew its utility when there was no breakthrough within the indemnity negotiations.
Moderna and J&J:
India did grant EUA to Moderna’s mRNA COVID-19 vaccine in June 2021, permitting Indian pharma firm Cipla to import vaccines. Nevertheless, in September, as home vaccine manufacturing picked up, the Centre determined to not import Moderna vaccines, in accordance with Reuters. The 2-dose vaccine additionally wants ultra-cold storage services which aren’t accessible in most Indian districts.
For the J&J vaccine, the Centre agreed to waive bridging medical research — research used for vaccines for which medical trials haven’t been carried out in India — and granted EUA in August 2021. J&J had agreed to supply the vaccine domestically with Organic E, throughout a extreme vaccine scarcity in September 2021. The Centre had been in talks to accumulate as many as 43.5 million doses of the single-dose Janssen vaccine; nevertheless, it later dropped these plans because the home manufacturing of indigenous vaccines picked up.
The Centre has allowed non-public corporations to accumulate these international jabs on their very own. Nevertheless, corporations have hesitated to position orders because of the availability of cheaper home vaccine choices.
Which different vaccines are nonetheless in pipeline?
As of date, about 97 per cent of India’s eligible inhabitants over 18 years has acquired their first COVID-19 vaccine shot whereas 80 per cent has acquired two doses.
Nevertheless, there are three vaccines nonetheless present process medical trials in India which can be used both for export functions or for vaccinating toddlers.
Bharat Biotech’s nasal vaccine (BBV154):
An intranasal adenoviral vaccine, BBV154 is at present in its third stage of human trials in 4 websites throughout India. This vaccine generates an immune response on the website of an infection (within the nasal space) and basically blocks each an infection and transmission of COVID-19. The only-dose vaccine might be administered to youngsters over the age of two with a non-invasive, needle-free method. The Centre is aiming to inoculate younger youngsters with the intranasal vaccine, to scale back transmission of the virus.
Gennova Biopharmaceutical’s Gemcovac:
India’s first mRNA COVID-19 vaccine — Gemcovac1-19 — is at present looking for EUA from the Centre. Cleared to stockpile over 21 lakh doses from the Central Medicine Laboratory (CDL), Kasauli, the Pune-based pharma firm has run into bother over allegations of violating medical trial protocol.
The Centre has alleged that the corporate didn’t adjust to medical trial protocol throughout Section-2 and three of human trials. Throughout these phases, the corporate had been cleared to check the neutralisation antibodies of the mRNA vaccine on volunteers on days 29, 43, 57, 119 and 209. Nevertheless, through the trial, day 57 was cancelled with out informing the Central Licencing Authority, the Centre says. The corporate has refuted these allegations and submitted all paperwork concerning the trials to the DGCI for approval.
Genique Lifesciences’ vaccine:
As per the Centre’s assertion to the Parliament, Gurugram-based Genique Lifesciences had began superior pre-clinical trials for its vaccine candidate in July 2021. Nevertheless, the vaccine has not but been advisable by ICMR for medical trials.
[ad_2]
Supply hyperlink









